Chemical Kinetics and Nuclear ChemistryHard
Question
The reaction 2N2O5(g) → 4NO2(g) + O2(g) follows first order kinetics. The pressure of a vessel containing only N2O5 was found to increase from 50 mm Hg to 87.5 mm Hg in 30 min. The pressure exerted by the gases after 60 min. will be (Assume temperature remains constant) :
Options
A.125 mm Hg
B.106.25 mm Hg
C.116.25 mm Hg
D.150 mm Hg
Solution
2N2O5(g) → 4NO2(g) + O2(g)
t = 0 50 0 0
t = 30 50 - 2x 4x x
⇒ 87.5 = 50 + 3x
⇒ 3x = 37.5 ⇒ x = 12.5
⇒ PN2O5 after 30 min = 50 - 25 = 25
⇒ t1/2 = 30 min.
Hence after 60 min, (two half lives), PN2O5 remaining =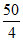 = 12.5 torr.
= 12.5 torr.
⇒ Hence decrease in PN2O5 = 50 - 12.5 = 37.5 torr.
⇒ PNO2 = 2 × 37.5 = 75 torr
PO2 =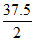 = 18.75 torr
= 18.75 torr
⇒ Ptotal = 12.5 + 75 + 18.75
= 106.25 torr.
t = 0 50 0 0
t = 30 50 - 2x 4x x
⇒ 87.5 = 50 + 3x
⇒ 3x = 37.5 ⇒ x = 12.5
⇒ PN2O5 after 30 min = 50 - 25 = 25
⇒ t1/2 = 30 min.
Hence after 60 min, (two half lives), PN2O5 remaining =
⇒ Hence decrease in PN2O5 = 50 - 12.5 = 37.5 torr.
⇒ PNO2 = 2 × 37.5 = 75 torr
PO2 =
⇒ Ptotal = 12.5 + 75 + 18.75
= 106.25 torr.
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
In the reaction : P + Q →R + S the time taken for 75% reaction of P is twice the time taken for 50% reaction of P. ...Consider the chemical reaction N2(g) + 3H2(g) → 2NH3(g). The rate of this reaction can be expressed in terms of time der...For the first-order reaction t99% = x × t90%. The value of ‘x’ will be...For a given reaction A → Product, rate is 1 × 10-4 M s-1 when [A] = 0.01 M and rate is 1.41 × 10-4 M s-1...The activation energy of a reaction can be determined from the slope of which of the following graphs ?...