JEE Advanced | 2013Chemical Kinetics and Nuclear ChemistryHard
Question
In the reaction :
P + Q →R + S
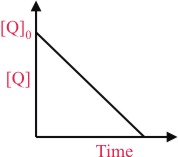
the time taken for 75% reaction of P is twice the time taken for 50% reaction of P. The concentrationof Q varies with reaction time as shown in the figure. The overall order of the reaction is -
Options
A.2
B.3
C.0
D.1
Solution
For reactant ′P′
Since t75% = 2 t50%
therefore order with respect to ′P′ is ′one′
For reactant ′Q′
linear variation of concentration of ′Q′ with time suggest that order with respect to ′Q′ is zero, therefore
overall order will be = 1 + 0 = 1
Correct Option (D)
(Assuming conc. of other reactant to be very large)
Since t75% = 2 t50%
therefore order with respect to ′P′ is ′one′
For reactant ′Q′
linear variation of concentration of ′Q′ with time suggest that order with respect to ′Q′ is zero, therefore
overall order will be = 1 + 0 = 1
Correct Option (D)
(Assuming conc. of other reactant to be very large)
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
For a certain reaction of order ‘n’, the time for half change t1/2 is given by $t_{1/2} = \frac{\left\lbrack 2 - \sqrt{2...For which of the following reactions K310/K300 would be maximum ?...If no catalyst (H+) is present in acid hydrolysis of ester (in above question) then rate constant K is :...Which of the following is an incorrect statement about first-order reaction?...The initial rate of hydrolysis of methyl acetate (1M) by a weak acid (HA, 1M) is 1/100th of that of a strong acid (HX, 1...