Chemical Kinetics and Nuclear ChemistryHard
Question
For a given reaction A → Product, rate is 1 × 10-4 M s-1 when [A] = 0.01 M and rate is 1.41 × 10-4 M s-1 when [A] = 0.02 M. Hence, rate law is :
Options
A.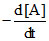 = k [A]2
= k [A]2
B.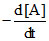 = k [A]
= k [A]
C.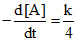 [A]
[A]
D.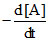 = k[A]1/2
= k[A]1/2
Solution
A → Product
We know, Rate = K [conc.]n
1 √ 10-4 = K [.01]n .....(i)
1.41 × 10-4 = K [.02]n .....(ii)
(i) / (ii)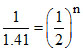
n =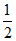
Then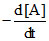 = K [A]1/2
= K [A]1/2
We know, Rate = K [conc.]n
1 √ 10-4 = K [.01]n .....(i)
1.41 × 10-4 = K [.02]n .....(ii)
(i) / (ii)
n =
Then
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
The rate of a chemical reaction doubles for every 10oC rise of temperature. If the temperature is raised by 50oC, the ra...The hydrolysis of an ester was carried out with 0.1 M H2SO4 and 0.1 M HCl separately. Which of the following expressions...The rate law for a reaction between the substances A and B is given by rate = K[A]n [B]m. On doubling the concentration ...Number of Faraday required to liberate 8 g. of H2 is :-...In a study of effect of temperature on reaction rate, the value of $\frac{1}{K}.\frac{dK}{dT}$ is found to be $\frac{1.2...