AIPMT | 2015Chemical Kinetics and Nuclear ChemistryHard
Question
The activation energy of a reaction can be determined from the slope of which of the following graphs ?
Options
A.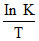 vs.T
vs.T
B.In K vs.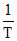
C.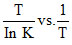
D.In K vs. T
Solution
Arrhenius equation
K = A.e−Ea/RT ⇒ lnK = lnA −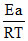
so, activation energy of reaction can be determined from the slope of lnK vs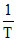
K = A.e−Ea/RT ⇒ lnK = lnA −
so, activation energy of reaction can be determined from the slope of lnK vs
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
The rate of a reaction A + 2B → P is 2 × 10−2 M/ min, when concentrations of each A and B are 1.0 M. If the rate of reac...For a first order reaction A → P, the temperature (T) dependent rate constant (k) was found to follow the equation...x mmol of KIO3 react completely with y mmol of KI to give I2 quantitavely. If z mmol of hypo are required for complete t...As the initial concentration increases from 0.75 to 1.55 M in a reaction, t1/2 decreases from 60 to 29 s. The order of t...How many coulomb of electricity would be required to reduce the iron in 32.9 g of potassium hexa cyano ferrate (III) K3F...