Atomic StructureHard
Question
Cv and Cp denote the molar specific heat capacities of a gas at constant volume and constant pressure, respectively. Then
(A) Cp - Cv is larger for a diatomic ideal gas than for a monoatomic ideal gas
(B) Cp + Cv is larger for a diatomic ideal gas than for a monoatomic ideal gas
(C) Cp/Cv is larger for a diatomic ideal gas than for a monatomic ideal gas
(D) Cp . Cv is larger for a diatomic ideal gas than for a monoatomic ideal gas
Which statements are correct :-
(A) Cp - Cv is larger for a diatomic ideal gas than for a monoatomic ideal gas
(B) Cp + Cv is larger for a diatomic ideal gas than for a monoatomic ideal gas
(C) Cp/Cv is larger for a diatomic ideal gas than for a monatomic ideal gas
(D) Cp . Cv is larger for a diatomic ideal gas than for a monoatomic ideal gas
Which statements are correct :-
Options
A.A & C
B.A, B & D
C.B & D
D.B, C & D
Solution
For monoatomic CV = 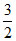 R, Cp =
R, Cp = 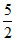 R
R
For diatomic CV = R, Cp =
R, Cp = 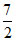 R
R
For diatomic CV =
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
In each fission of 92U235 releases 200 MeV, how many fission must occur per second to pruduce power of kW ?...Nuclear reactions are given as -(i) ⎕ (n, p)15p32(ii) ⎕ (p, α)8O16(iii) 7N14 (⎕, p) 6C14 missing ...We wish to seen inside an atom. Assuming the atom to have a diameter of 10 pm, thismeans that one must be able to resolv...Following are graphs of electric field variation with distance (r) for four different charge configurations.Choose the c...Suppose an electron is attracted towards the origin by a force k/r where ′k′ is a constant and ′r̸...