Atomic StructureHard
Question
Suppose an electron is attracted towards the origin by a force k/r where ′k′ is a constant and ′r′ is the distance of the electron from the origin. By applying Bohr model to this system, the radius of the nth orbital of the electron is found to be ′rn′ and the kinetic energy of the electron to be Tn. Then which of the following is true?
Options
A.Tn ∝ 1/n2, rn ∝ n2
B.Tn independent of n, rn ∝ n
C.Tn ∝ 1/n, rn ∝ n
D.Tn ∝ 1/n, rn ∝ n2
Solution
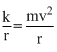
mv2 = k
n
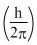 = mvr ⇒ r ∝ n and T =
= mvr ⇒ r ∝ n and T = 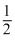 mv2 is independent of n.
mv2 is independent of n.Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
Which of the following statement(s) is (are) correct ?...he decay constant of radioactive sample is λ. The half-life and mean-life of the sample are respectively given by...Consider 3rd orbit of He+ (Helium), using non-relativistic approach, the speed of electron in this orbit will be [given ...Out of the following which one is not a possible energy for a photon to be emitted by hydrogen atom according to Bohr...Photoelectric effect supports quantum nature of light because...