Chemical Kinetics and Nuclear ChemistryHard
Question
Rates of hydration of the following alkenes are :-
(P) CH3-O-CH = CH2
(Q) F-CH = CH2
(R) CH3 -CH = CH2
(S)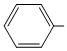 CH = CH2
CH = CH2
(P) CH3-O-CH = CH2
(Q) F-CH = CH2
(R) CH3 -CH = CH2
(S)
 CH = CH2
CH = CH2Options
A.P > Q > R > S
B.S > R > Q > P
C.P > S > R > Q
D.R > S > P > Q
Solution
Reactivity towards hydration ∝ stability of carbocation
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
Which of the following molecules is most suitable to disperse benzene in water ?...The half-life of a radioactive isotope is three hours. If the initial mass of the isotope were 256 g, the mass of it rem...For a reaction A → 2B, rate of disappearance of ′A′ is related to the rate of appearance of ′B&#...The composition of N2O5 is a first order reaction represented by: N2O5 → N2O4 + 1/2O2.After 20 min the volume of O...Kyoto Protocol was endorsed at :...