Chemical Kinetics and Nuclear ChemistryHard
Question
The composition of N2O5 is a first order reaction represented by: N2O5 → N2O4 + 1/2O2.After 20 min the volume of O2 produced is 10 mL and at the end of the reaction 40 mL. The rate constant is equal to
Options
A.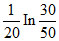
B.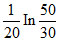
C.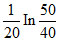
D.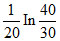
Topic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
For the reaction A → Products (order = n), the theoretical time of completion of reaction is...x mmol of KIO3 react completely with y mmol of KI to give I2 quantitavely. If z mmol of hypo are required for complete t...The hydrolysis of an ester was carried out with 0.1 M H2SO4 and 0.1 M HCl separately. Which of the following expressions...A radioactive substance does not emit:...For a given reaction A → Product, rate is 1 × 10-4 M s-1 when [A] = 0.01 M and rate is 1.41 × 10-4 M s-1...