Chemical Kinetics and Nuclear ChemistryHard
Question
Which of the following molecules is most suitable to disperse benzene in water ?
Options
A.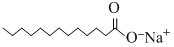

B.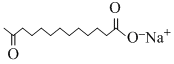

C.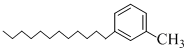

D.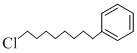

Solution
Benzene is non-polar and hence dissolves non-polar compounds only (c) is non-polarhence it dissolves in benzene.
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
Determine the average life of U238 having t0.5 = 138.5 day....On increasing the temperature, the half-life of reactions...Arrange the following in order of decreasing boiling point :-(I) n-butane (II) n-butanol (III) n-butyl chloride (IV) Iso...The rate law for the dimerisation of NO2 is = k [NO2]2which of the following changes will change the value of the specif...The following data certain to reaction between A and B ; Which of the following inference(s) can be drawn from the above...