Chemical Kinetics and Nuclear ChemistryHard
Question
The half-life of a radioactive isotope is three hours. If the initial mass of the isotope were 256 g, the mass of it remaining undecayed after 18 hours would be
Options
A.8.0 g
B.12.0 g
C.16.0 g
D.4.0 g
Solution
t1/2 = 3 hrs. Initial mass (C0) = 256 g
∴ Cp =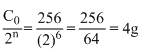
∴ Cp =
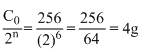
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
For the first-order reaction Tav (average life), T50 and T75 in the increasing order are :...A Catalyst...Higher order (>3) reactions are rare due to : -...The activation energy of a reaction can be determined from the slope of which of the following graphs ?...For the reversible reaction A$\rightleftharpoons$B, the time at which the rate of change in concentration of ‘A’ is equa...