Atomic StructureHard
Question
When photo energy 4.25 eV strike the surface of a metal A, the ejected photoelectrons have maximum kinetic energy TA expressed in eV and de-Broglie wavelength λA. The maximum kinetic energy of photoelectrons liberated from another metal B by photons of energy 4.70 eV is TB = 2λA, then
Options
A.the work function of A is 2.25 eV
B.the work function of B is 4.20 eV
C.TA = 2.00 eV
D.TB = 2.75 eV
Solution
Kmax = E - W
Therefore, .....(i)
TA = 4.25 - WB .....(ii)
From Eqs. (i) and (ii),
WB - WWA = 1.95 eV .....(iii)
de-Broglie wavelength is given
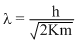 or
or 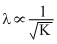 K = KE of electron
K = KE of electron
∴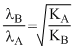
or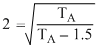
This gives, TA = 2 eV
From Eq. (i) WB = WA + 1.95 eV = (2.25 + 1.95)eV
or TB = 4.70 - WB = 4.70 - 4.20 = 0.50 eV
Therefore, .....(i)
TA = 4.25 - WB .....(ii)
From Eqs. (i) and (ii),
WB - WWA = 1.95 eV .....(iii)
de-Broglie wavelength is given
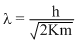 or
or 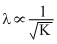 K = KE of electron
K = KE of electron ∴
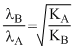
or
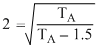
This gives, TA = 2 eV
From Eq. (i) WB = WA + 1.95 eV = (2.25 + 1.95)eV
or TB = 4.70 - WB = 4.70 - 4.20 = 0.50 eV
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
In an n-p-n transistor circuit, the collector current is 10 mA. If 90 % of the electrons emitted reach the collector...A gas is compressed isothermally to half its initial volume. The same gas is compressed separately throughan adiabatic p...The X-ray beam coming from an X-ray tube will be...The radius of the orbit of an electron in a Hydrogen-like atom is 4.5 a0, where a0 is the Bohr radius. Its orbital angul...Light of wavelength 4000 is incident on a metal plate whose work function is 2eV.What is maximum kinetic energy of emitt...