JEE Advanced | 2013Atomic StructureHard
Question
The radius of the orbit of an electron in a Hydrogen-like atom is 4.5 a0, where a0 is the Bohr radius. Its orbital angular momentum is 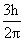 It is given that h is Planck constant and R is Rydberg constant. The possible wavelength (s), when the atom de-excites, is (are) :-
It is given that h is Planck constant and R is Rydberg constant. The possible wavelength (s), when the atom de-excites, is (are) :-
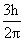 It is given that h is Planck constant and R is Rydberg constant. The possible wavelength (s), when the atom de-excites, is (are) :-
It is given that h is Planck constant and R is Rydberg constant. The possible wavelength (s), when the atom de-excites, is (are) :-Options
A.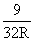
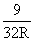
B.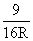
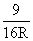
C.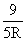
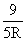
D.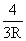
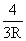
Solution
Angular momentum 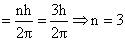
Also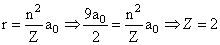
for de-excitation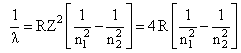
for n = 3 to n = 1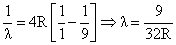
for n = 3 to n = 2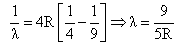
for n = 2 to n = 1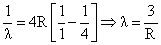
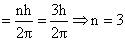
Also
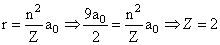
for de-excitation
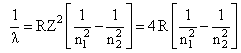
for n = 3 to n = 1
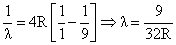
for n = 3 to n = 2
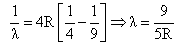
for n = 2 to n = 1
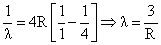
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
Solid targets of different elements are bombarded by highly energetic electron beam. the frequency (f) of the characteri...Two electrons are moving in orbits of two hydrogen like atoms with speeds $3 \times 10^{5}\text{ }m/s$ and $2.5 \times 1...The kinetic in the potential different of 100V,...Two thin conducting shells of radii R and 2R are shown in figure. The outer shell carries a charge +Q and inner shell is...When photo energy 4.25 eV strike the surface of a metal A, the ejected photoelectrons have maximum kinetic energy TA exp...