Atomic StructureHard
Question
Two cylinders A and B fitted with pistons contain equal amounts of an ideal diatomic gas at 300 K. The piston of A is free to move while that of B is held fixed. The same amount of heat is given to the gas in each cylinder. If the rise in temperature of the gas in A is 30K, then the rise in temperature of the gas in B is :-
Options
A.30 K
B.18 K
C.50 K
D.42 K
Solution
In both cylinders A and B the gases are diatomic (γ = 1.4). Piston A is free to move i.e., it is isobaric process. Piston B is fixed i.e., it is isochoric process. If same amount of heat ᐃQ is given to both then
(ᐃQ)isobaric = (ᐃQ)isochoric ⇒ μCp(ᐃT)A = μCv(ᐃT)B
⇒ (ᐃT)B =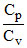 (ᐃT)A = γ (ᐃT)A = 1.4 × 30 = 42 K
(ᐃT)A = γ (ᐃT)A = 1.4 × 30 = 42 K
(ᐃQ)isobaric = (ᐃQ)isochoric ⇒ μCp(ᐃT)A = μCv(ᐃT)B
⇒ (ᐃT)B =
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
In the Bohr model of a hydrogen atom, the centripetal force is furnished by the coulomb attraction between the proton an...Two hydrogen atoms are in excited state with electrons residing in n = 2. First one is moving towards left and emits a p...The diagram shows the energy levels for an electron in a certain atom. Which transition shown represents the emission of...In Bohr′s theory. relation between principal quantum number n and radius of orbit r is :...Which of the following has the highest neutrons ratio ?...