Atomic StructureHard
Question
In the Bohr model of a hydrogen atom, the centripetal force is furnished by the coulomb attraction between the proton and the electron. If a0 is the radius of the ground state orbit, m is the mass, e is the charge on the electron and λ0 is the vacuum permittivity, the speed of the electron in first excited state is :-
Options
A.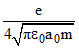
B.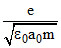
C.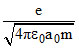
D.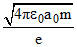
Solution
In ground state,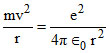
v =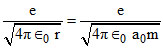
∵ v ∝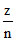
∴ velocity in first excited state (n = 2)
v2 =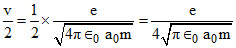
v =
∵ v ∝
∴ velocity in first excited state (n = 2)
v2 =
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
The transition from the state n = 3 to n = 1 in a hydrogen like atom results in ultraviolet radiation.Infrared radiation...A radioactive material has half-life of 10 days. what fraction of the material would remain after 30 days?...The work function of a substance is $4.0$ eV. The longest wavelength of light that can cause photoelectron emission from...The speed of an electron having a wavelength of 10-10 m is...In a hydrogen like atom in which every atom is in a particular excited state. Now a stream of photons of energy E0 bomba...