Atomic StructureHard
Question
Which of the following has the highest neutrons ratio ?
Options
A.92U235
B.8O16
C.2He4
D.26Fe56
Solution
Neutrons ratio that is 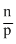 determines the stability of nucleus. (Here, n is number of neutrons and p is protons).
determines the stability of nucleus. (Here, n is number of neutrons and p is protons).
Now, n = 235 - 92 = 143
p = 92
For 92U235; Neutron ratio =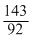 = 1.55
= 1.55
Which is the highest no. So, 92U235; is most unstable.
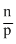 determines the stability of nucleus. (Here, n is number of neutrons and p is protons).
determines the stability of nucleus. (Here, n is number of neutrons and p is protons).Now, n = 235 - 92 = 143
p = 92
For 92U235; Neutron ratio =
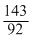 = 1.55
= 1.55Which is the highest no. So, 92U235; is most unstable.
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
The radius of the orbit of an electron in a Hydrogen-like atom is 4.5 a0, where a0 is the Bohr radius. Its orbital angul...If the binding energy per nucleon of a nuclide is high then...The wavelength of the first spectral line in the Balmer series of hydrogen atom is 6561 The wavelength of the second spe...The activity of radioactive sample is measured as 9750 counts per minute at t = 0 and as 975 counts per minute at t = 5 ...The ground state energy of hydrogen atom is -13.6eV. What is the potential energy of the electron in this state?...