Chemical Kinetics and Nuclear ChemistryHard
Question
For the complex reaction A 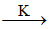 product
product
Ea1 = 180 kJ/mole Ea2 = 80 kJ/mole
Ea3 = 50 kJ/mole
Over all rate constant K is related to individual rate constant by the equation K =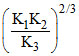
Activation energy (KJ/mol) for the overall reaction is ?
Ea1 = 180 kJ/mole Ea2 = 80 kJ/mole
Ea3 = 50 kJ/mole
Over all rate constant K is related to individual rate constant by the equation K =
Activation energy (KJ/mol) for the overall reaction is ?
Options
A.150
B.140
C.100
D.None of these
Solution
Ea = 2/3 [Ea1 + Ea2 - Ea3]
Ea = 2/3 [180 + 80 - 50]
Ea = 140
Ea = 2/3 [180 + 80 - 50]
Ea = 140
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
For reaction aA → xP, when [A] = 2.2 mM, the rate was found to be 2.4mMs-. On reducing concentration of half, the ...The reaction A → B follows first order kinetics. The time taken for 0.8 mol of A to produce 0.6 mol of B is 1 hr. ...Zn + 2 H+ → Zn2+ + H2Half-life period is independent of concentration of zinc at constant pH. For the constant con...In terms of polar character, which of the following order is correct :-...emits 8α - particles and 6β - particles. The netron /proton ratio in the product nucleus is :...