Chemical Kinetics and Nuclear ChemistryHard
Question
The reaction A → B follows first order kinetics. The time taken for 0.8 mol of A to produce 0.6 mol of B is 1 hr. What is the time taken for the conversion of 0.9 mol of A to product 0.675 mol of B ?
Options
A.1 hr
B.0.5 hr
C.0.25 hr
D.2 hr
Solution
Time for the conversion of 0.8 mol of A to 0.6 mol of
B =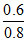 = 0.75 = t75%
= 0.75 = t75%
Similarly, time for the conversion of 0.9 mol of A to 0.675 mol of
B =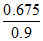 = 0.75 = t75%
= 0.75 = t75%
Hence, t75% in both case = 1 hr
B =
Similarly, time for the conversion of 0.9 mol of A to 0.675 mol of
B =
Hence, t75% in both case = 1 hr
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
The rate of a reaction increases four-fold when the concentration of reactant is increased 16 times. If the rate of reac...The physical adsorption of gases on the solid surface is due to:...Consider the following first-order decomposition reaction. A4(g)→ 4A(g)Which of the following statement(s) is/are correc...Number of natural life times (Tav) required for a first-order reaction to achieve 99.9% level of completion is :...6C12 and 1T3 are formed in nature due to the nuclear reaction of neutron with...