Chemical Kinetics and Nuclear ChemistryHard
Question
For reaction aA → xP, when [A] = 2.2 mM, the rate was found to be 2.4mMs-. On reducing concentration of half, the rate changes to 0.6mMs- -. The order of reaction with reaspect to A is
Options
A.1.5
B.2.0
C.2.5
D.3.0
Solution
When the concentration of reactant is reduced to half its initial value, the rate is rediced half its initial value, the rate is reduced by 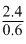 = times
= times
It means, rate ∝ [reactant]2
So, order of reaction = 2
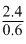 = times
= timesIt means, rate ∝ [reactant]2
So, order of reaction = 2
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
For the first-order parallel reactions, starting with 1 M of A, which of the following statement(s) is/are correct? $A\o...The reaction 2N2O5(g) → 4NO2(g) + O2(g) follows first order kinetics. The pressure of a vessel containing only N2O...A(g) → B(g) + (α - particles)(α-particles are helium nuclei, so will form helium gas by trapping electro...A second order reaction requires 70 min to change the concentration of reactants from 0.08 M to 0.01 M. How much time wi...The activation energies of two reactions are E1 and E2 with E1 > E2. If the temperature of the reacting system is inc...