Chemical Kinetics and Nuclear ChemistryHard
Question
Zn + 2 H+ → Zn2+ + H2
Half-life period is independent of concentration of zinc at constant pH. For the constant concentration of Zn, rate becomes 100 times when pH is decreased from 3 to 2. Hence ,
Half-life period is independent of concentration of zinc at constant pH. For the constant concentration of Zn, rate becomes 100 times when pH is decreased from 3 to 2. Hence ,
Options
A.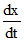 = k [Zn]0[H+]2
= k [Zn]0[H+]2
B.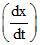 = k [Zn][H+]2
= k [Zn][H+]2
C.rate is not affected if concentration of zinc is made four times and that of H+ ion is halved
D.rate becomes four times if concentration of H+ ion is doubled at constant Zn concentration
Solution
(B) Half life period independent of conc. of Zn than order W.R.T. Zn is 1st
Rate = k [Zn][H+]n we can write
by data
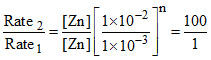
Given n = 2
Then Rate = k [Zn][H+]2
(C) Conc. of Zn four times and H+ ion is halved
Rate = k[4 × Zn]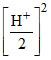
Rate = k [Zn][H+]2
Rate remians and same.
(D) Similarly if conc. of H+ doubled and [Zn] constant
Then Rate2 = k [Zn] [2H+]2
Rate2 = 4 k [Zn] [H+]2
Rate2 = 4 x Rate1
Rate = k [Zn][H+]n we can write
by data
Given n = 2
Then Rate = k [Zn][H+]2
(C) Conc. of Zn four times and H+ ion is halved
Rate = k[4 × Zn]
Rate = k [Zn][H+]2
Rate remians and same.
(D) Similarly if conc. of H+ doubled and [Zn] constant
Then Rate2 = k [Zn] [2H+]2
Rate2 = 4 k [Zn] [H+]2
Rate2 = 4 x Rate1
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
The compound A decomposed (A → product) by first order kinetics. If the concentration of A dropped from 0.1 M to 0...Graph between log k and 1/T (k is rate constant in s-1 and T is the temperature in K) is a straight line. As shown in fi...For the non-equilibrium process, A + B → products, the rate is first-order w.r.t. A and second-order w.r.t. B. If ...With the help of following reactions, arrange metals A,B,D and E in decreasing order of their reactivity :-(i) B + ANO3 ...Which of the following statements are correct ?...