Chemical Kinetics and Nuclear ChemistryHard
Question
For a process A+ B Products, the rate is first order with respect to A and First order with respect to B. When 1.0 mol each of A and B are taken in one litre vessel, the initial rate is 1 × 10-2 mol L-1 S-1. The rate of reaction when 90% of the reactant have been converted into products would be :-
Options
A.1 × 10-2 mol L-1 S-1
B.1 × 10-4 mol L-1 S-1
C.1 × 10-3 mol L-1 S-1
D.1 × 10-5 mol L-1 S-1
Solution
r = K [A]1 [B]1
∴ r1 = K (1)1 (1)1
and r2 = K (0.1)1 (0.1)1
⇒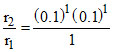
⇒ r2 = 10-2 × 10-2 = 10-4 mol L-1 s-1
∴ r1 = K (1)1 (1)1
and r2 = K (0.1)1 (0.1)1
⇒
⇒ r2 = 10-2 × 10-2 = 10-4 mol L-1 s-1
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
Consider the following statements. The law for the acid-catalysed hydrolysis of an ester being given as rate = K [H+] [e...The rate law for the dimerisation of NO2 is = k [NO2]2which of the following changes will change the value of the specif...The compound A decomposed (A → product) by first order kinetics. If the concentration of A dropped from 0.1 M to 0...The hydrolysis of an ester was carried out with 0.1 M H2SO4 and 0.1 M HCl separately. Which of the following expressions...Consider the chemical reaction N2(g) + 3H2(g) → 2NH3(g). The rate of this reaction can be expressed in terms of time der...