Chemical Kinetics and Nuclear ChemistryHard
Question
The hydrolysis of an ester was carried out with 0.1 M H2SO4 and 0.1 M HCl separately. Which of the following expressions between the rate constants is expected ? The rate expression being rate = k[H+][ester]
Options
A.kHCl = kH2SO4
B.kHCl > kH2SO4
C.kHCl < kH2SO4
D.kH2SO4 = kHCl
Solution
[H2SO4] = 0.1 M = 0.1 × 2 = 0.2 N
[HCl] = 0.1 N
In case of H2SO4
r1 = k[H+] [Ester]
kH2SO4 =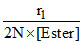
In case of HCl
r2 = k[H+] [Ester]
kHCl =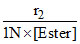
Hence kHCl > kH2SO4
[HCl] = 0.1 N
In case of H2SO4
r1 = k[H+] [Ester]
kH2SO4 =
In case of HCl
r2 = k[H+] [Ester]
kHCl =
Hence kHCl > kH2SO4
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
The correct order of N-compounds in its decreasing order of oxidation states is...At 370 K a gaseous reaction X → 2Y + Z is found to be first order. Starting the reaction from pure X the total pre...Incorrect order of solubility is :...For the non-stoichiometre reaction 2A + B → C + D, the following kinetic data were obtained in three separate expe...When ethyl acetate was hydrolyzed in the presence of 0.1 m HCl, the constant was found to be 5.40 × 10-5 s-1. But w...