NEETChemical Kinetics and Nuclear ChemistryHard
Question
The rate law for the dimerisation of NO2 is
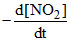 = k [NO2]2
= k [NO2]2
which of the following changes will change the value of the specific rate constant , k :
which of the following changes will change the value of the specific rate constant , k :
Options
A.Doubling the total pressure on the system
B.Doubling the temperature
C.Both of (A) and (B)
D.None of the above
Solution
Rate constant change on changing temperature .
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
The addition of a catalyst during a chemical reaction alters which of the following quantities?...A reaction is catalysed by H+ ion;and in the rate law the dependence of rate is of first order with respect to the conce...Purple of cassius is colloidal solution of :...A radioactive element has a half life of one day. After three days the amount of the element left will be :...If = k [H+]n and rate becomes 100 times when pH changes from 2 to 1. Hence, order is :...