ThermodynamicsHard
Question
A system is taken along the paths A and B as shown. If the amounts of heat given in these processes are ᐃQA and ᐃQB respectively then :-
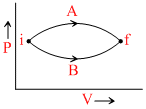

Options
A.ᐃQA = ᐃQB
B.ᐃQA ≥ ᐃQB
C.ᐃQA < ᐃQB
D.ᐃQA > ᐃQB
More Thermodynamics Questions
A Carnot engine, whose efficiency is 40% takesin heat from a source maintained at atemperature of 500 K. It is desired t...The figure shows the P-V plot of an ideal gas taken through a cycle ABCDA. The part ABC is a semi-circle and CDA is half...5.6 liter of helium gas at STP is adiabatically compressed to 0.7 lit. Taking the initial temperaure to be T1, the work ...An ideal gas goes from state A to state B via three different processes as indicated in the P-V diagramIf Q1, Q2, Q3 ind...One mole of a monatomic ideal gas is taken along two cyclic processes E → F → G → E and E → F &#...