ThermodynamicsHard
Question
One mole of a monatomic ideal gas is taken along two cyclic processes E → F → G → E and E → F → H → E as shown in the PV diagram. The processes involved are purely isochoric, isobaric, isothermal or adiabatic.
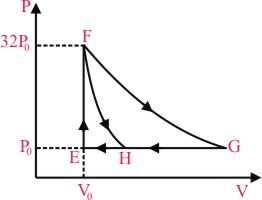
Match the paths in List I with the magnitudes of the work done in the List II and select the correct answer using the codes given blow the lists.


Match the paths in List I with the magnitudes of the work done in the List II and select the correct answer using the codes given blow the lists.
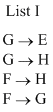

Options
A.P → 4
Q → 3
R → 2
S → 1
Q → 3
R → 2
S → 1
B.P → 4
Q → 3
R → 1
S → 2
Q → 3
R → 1
S → 2
C.P → 3
Q → 1
R → 2
S → 4
Q → 1
R → 2
S → 4
D.P → 1
Q → 3
R → 2
S → 4
Q → 3
R → 2
S → 4
Solution
(P) Process GE is isobaric
So work done = P |ᐃV| = P0 |(VG - VE)|
= P0 |(32V0 - V0)|
= 31V0V0
(Q) Process GH is isobaric so
So work done = P0 |(32V0 - 8V0)|
= 24 P0V0
(R) Since process FH is adiabatic so 32 P0V05/3 = P0VH5/3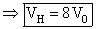
& work done W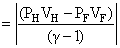
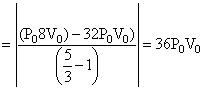
(S) Process FG is isothermal
so work done = nRT ln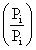
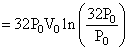
= 160 P0V0 ln2.
So work done = P |ᐃV| = P0 |(VG - VE)|
= P0 |(32V0 - V0)|
= 31V0V0
(Q) Process GH is isobaric so
So work done = P0 |(32V0 - 8V0)|
= 24 P0V0
(R) Since process FH is adiabatic so 32 P0V05/3 = P0VH5/3
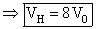
& work done W
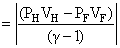
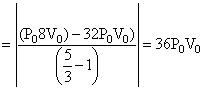
(S) Process FG is isothermal
so work done = nRT ln
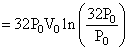
= 160 P0V0 ln2.
Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
The intensity of radiation emitted by the sun has its maximum value at a wavelength of 510 nm and emitted by the north s...At room temperature, the rms speed of the molecules of a certain diatomic gas is found to be 1930 m/s. The gas is...In an adiabatic change, the pressure and temperature of a monoatomic gas are related with relation as P ∝ TC, wher...When a solid is converted into a gas, directly by heating then this process is known as :...A black body is heated from 27oC to 127oC. The ratio of their energies of radiation emitted will be:...