ThermodynamicsHard
Question
An ideal gas goes from state A to state B via three different processes as indicated in the P-V diagramIf Q1, Q2, Q3 indicate the heat absorbed by the gas along the three processes and ᐃU1, ᐃU2, ᐃU3 indicate the change in internal energy along the three processes respectively, then
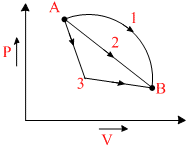

Options
A.Q3 > Q2 > Q1 and ᐃU1 > ᐃU2 > ᐃU3
B.Q1 > Q2 > Q3 and ᐃU1 = ᐃU2 = ᐃU3
C.Q3 > Q2 > Q1 and ᐃU1 = ᐃU2 = ᐃU3
D.Q1 = Q2 = Q3 and ᐃU1 > ᐃU2 > ᐃU3
Solution
ᐃQ = ᐃU + ᐃW
ᐃU is same Q1 > Q2 > Q3
ᐃU is same Q1 > Q2 > Q3
Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
Amount of work which can be obtained from 200 cal heat, will be :...A solid body of constant heat capacity 1 Jo/C is being heated by keeping it in contact with reservoirs in two ways -(i) ...The unit of gas constant R is :-...An ideal gas is initially at temperature T and volume V. Its volume is increased by ᐃV due to an increase in tempe...The process in which the heat given to a system is completely transformed into work is for ddeal gas : -...