ThermodynamicsHardBloom L4
Question
The figure shows the P-V plot of an ideal gas taken through a cycle ABCDA. The part ABC is a semi-circle and CDA is half of an ellipse. Then,
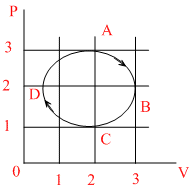

Options
A.the process during the path A → B is isothermal
B.heat flows out of the gas during the path B → C → D
C.work done during the path A → B → C is zero
D.positive work is done by the gas in the cycle ABCDA
Solution
ᐃQ = ᐃU + w
For process B → C → D
ᐃU is negative as well as W is also negative
For process B → C → D
ᐃU is negative as well as W is also negative
Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
An ideal gas goes from state A to state B via three different processes as indicated in the P-V diagramIf Q1, Q2, Q3 ind...At a constant temperature and volume for a given gas, pressure...The efficienty of a Carnot engine operating with reservoir tempeture of 100oC and −23oC will be :...A bullet moving with velocity v collides against wall. consequently half of its kinetic energy is converted into heat. I...For an ideal gas...