Gaseous StateHard
Question
Calculate the entropy change involved in the isothermal reversible expansion of 5 mol of ideal gas from a volume of 10L to 100 L at 300 K :-
Options
A.191.47 JK-1
B.95.7 JK-1
C.41.57 JK-1
D.83.14 JK-1
Solution
ᐃS = nR ln 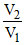
= 2.303 × 5 × 8.314 log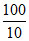 = 95.7JK-1
= 95.7JK-1
= 2.303 × 5 × 8.314 log
Create a free account to view solution
View Solution FreeMore Gaseous State Questions
Henry′s law constants for four gases are given below at 293 K. Which one is more soluble in water at that temperat...The curve of pressure volume (PV) against pressure (P) of the gas at a particular temperature is as shown, according to ...What volume will be occupied by a sample of gas at 40oC and 720 mm μg if its volume at 20oC and 750 mm μg was ...A gas cylinder containing cooling gas can withstand a pressure of 14.9 atmosphere. The pressure gauge of cylinder indica...Potassium hydroxide solutions are used to absorb CO2. How many litres of CO2 at 1.00 atm and 22oC would be absorbed by a...