Gaseous StateHard
Question
The curve of pressure volume (PV) against pressure (P) of the gas at a particular temperature is as shown, according to the graph which of the following is incorrect (in the low pressure region):
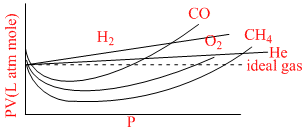

Options
A.H2 and He shows +ve deviation from ideal gas equation.
B.CO, CH4 and O2 show negative deviation from ideal gas equation.
C.H2 and He show negative deviation while CO2, CH4 and O2 show positive deviation.
D.H2 and He are less compressible than that of an ideal gas while CO2, CH4 and O2 more compressible than that of ideal gas.
Solution
If Z > 1 positive deviation
Z < 1 negative deviation
Z < 1 negative deviation
Create a free account to view solution
View Solution FreeMore Gaseous State Questions
Which of the following is not an assumption of the kinetic theory of gases ?...Which of the following expression correctly represents the relationship between the average kinetic energy of CO and N2 ...Which of the following combination will produce H2 gas?...A gaseous mixture was prepared by takig equal moles of CO and N2. If the total pressure of the mixture was found 1 atmos...By what factor does the average velocity of a gaseous molecule increase when the temperature (in Kelvin) is doubled ?...