Gaseous StateHard
Question
Potassium hydroxide solutions are used to absorb CO2. How many litres of CO2 at 1.00 atm and 22oC would be absorbed by an aqueous solution containing 15.0 g of KOH ? (Take R = 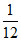 l atm / K/mole)
l atm / K/mole)
2KOH + CO2 → K2CO3 + H2O
2KOH + CO2 → K2CO3 + H2O
Options
A.3.24 L
B.1.62 L
C.6.48 L
D.0.324 L
More Gaseous State Questions
A hypothetical gaseous element having molecular formula Mx may be changed to another gaseous allotrope having molecular ...A liquid is in equilibrium with its vapour at it′s boiling point. On the average, the molecules in the two phases ...The correct order of normal boiling points of O2, N2, NH3 and CH4, for whom the values of vander Waal′s constant &...At what temperature, the average speed of gas molecules be double of that at temperature, 27oC?...Two glass bulbs A and B are connected by a very small tube having a stop cock. Bulb A has a volume of 100 cm3 and contai...