Gaseous StateHard
Question
A gas cylinder containing cooling gas can withstand a pressure of 14.9 atmosphere. The pressure gauge of cylinder indicates 12 atmosphere gauge of cylinder indicates 12 atmosphere at 27oC. Due to sudden fire in the building the temperature starts rusting. The temperature at which cylinder explodes is :
Options
A.87.5oC
B.99.5oC
C.115.5oC
D.135.5oC
Solution
From Charle′s law
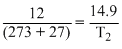
T2 =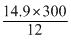 = 372.5 K
= 372.5 K
= 372.5 - 273 = 99.5oC
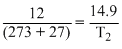
T2 =
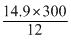 = 372.5 K
= 372.5 K= 372.5 - 273 = 99.5oC
Create a free account to view solution
View Solution FreeMore Gaseous State Questions
For the non-zero values of force of attraction between gas molecules, gas equation will be :...Which is not correct about the Schlocky defects?...Equal masses of methane and oxygen are mixed in an empty container at 25oC. The fraction of the total pressure exerted b...Consider the following reactions in which all the reactants are products are in gaseous state :- 2PQ ⇋ P2 + Q2 ; K...A gaseous alkane is exploded with oxygen. The volume of O2 for complete combustion to CO2 formed is in the ratio 7/4. Th...