Gaseous StateHard
Question
What volume will be occupied by a sample of gas at 40oC and 720 mm μg if its volume at 20oC and 750 mm μg was 250 mL?
Options
A.278.19 mL
B.378.19 mL
C.400 mL
D.187 mL
Solution
VC = 3b = 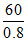 = 75cm3 mol-1
= 75cm3 mol-1
⇒ b = 25 cm3 mol-1 = 0.025 L mol-1
∴ TC =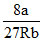 ⇒ a = 3.375
⇒ a = 3.375
⇒ b = 25 cm3 mol-1 = 0.025 L mol-1
∴ TC =
Create a free account to view solution
View Solution FreeMore Gaseous State Questions
A certain gas takes three times as long to effuse out as helium. Its molecular mass will be:...What is the total pressure exerted by the mixture of 7.0 g of N2, 2g of hydrogen and 8.0 g of sulphur dioxide gases in a...Just below this temperature, gases would theoretically have a negative volume (which is however not possible)-...N2 + 3H2 → 2NH3. 1 mol N2 and 4 mol H2 are taken in 15 L flask at 27oC. After complete conversion of N2 into NH3, ...SO2 at STP contained in a flask was replaced by O2 under identical conditions of pressure, temperature and volume. Then ...