Chemical EquilibriumHard
Question
For the reversible reaction, N2(g) + 3H2(g) ⇋ 2NH3 at 500oC, the value of Kp is 1.44 × 10-5 when partial pressure is measured in atmosphere. The corresponsing value of Kc with concentration in mol / litre is :
Options
A.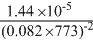
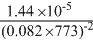
B.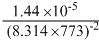
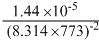
C.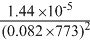
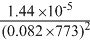
D.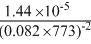
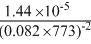
Solution
N2(g) + 3H2(g) ⇋ 2NH3(g)
Kp = Kc(RT)ᐃn
ᐃn = 2 - (1 + 3) = - 2
∴ Kc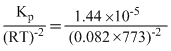
(T = 500 + 273 = 773K)
Kp = Kc(RT)ᐃn
ᐃn = 2 - (1 + 3) = - 2
∴ Kc
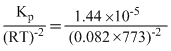
(T = 500 + 273 = 773K)
Create a free account to view solution
View Solution FreeMore Chemical Equilibrium Questions
In a 5.76 L vessel, 0.5 moles of H2 gas and 0.5 moles of I2 vapours are allowed to react to form HI (g) at 447°C, then t...When S in the form of S8 is heated at 900 K, the initial pressure of 1 atm falls by 30% at equilibrium. This is because ...XY2 dissociates as :XY2(g) ⇋ XY(g) + Y(g)initial pressure of XY2 is 600 mm Hg. The total pressure at equilibrium i...The degree of dissociation of PCl5 (a) obeying the equilibrium, PCl5 ⇋ PCl3 + Cl2, is approximately related to the...At 1000oC and a pressure of 16 atm, the equilibrium constant of the reaction CO2(g) + C(s) $\rightleftharpoons$2CO(g) is...