Chemical EquilibriumHard
Question
XY2 dissociates as :
XY2(g) ⇋ XY(g) + Y(g)
initial pressure of XY2 is 600 mm Hg. The total pressure at equilibrium is 800 mm Hg. Assuming volume of system to remain constant, the value of Kp is :-
XY2(g) ⇋ XY(g) + Y(g)
initial pressure of XY2 is 600 mm Hg. The total pressure at equilibrium is 800 mm Hg. Assuming volume of system to remain constant, the value of Kp is :-
Options
A.50
B.100
C.200
D.400
Solution
XY2(g) ⇋ XY(g) + Y(g)
600 0 0 Initially
600-P P P at equilibrium
given 600 - P + P + P = 800
P = 200 mm of Hg
KP =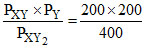 = 100
= 100
600 0 0 Initially
600-P P P at equilibrium
given 600 - P + P + P = 800
P = 200 mm of Hg
KP =
Create a free account to view solution
View Solution FreeMore Chemical Equilibrium Questions
At 0°C and 1 atm pressure, 1 L of N2O4 decomposes to NO2 according to the equation N2O4(g) $\rightleftharpoons$2NO2 (g)....The activation energies for the forward and reverse elementary reactions in the system A$\rightleftharpoons$B are 10.303...What is the equilibrium expression for the reaction P4(s) + 5O2(g) ⇋ P4O10(s) ?...The dissociation of ammonium carbamate may be represented by the equation NH4CO2NH2(s) ⇋ 2NH3(g) + CO2(g)ᐃH0...The degree of dissociation of SO3 is α at equilibrium pressure P0. Kp for 2SO3(g) ⇋ 2SO2(g) + O2(g) is...