Mole ConceptHard
Question
When Cl2 gas reacts with hot and concentrated sodium hydroxide solution, the oxidation number of chlorine changes from
Options
A.zero to +1 and zero to -5
B. zero to -1 and zero to +5
C.zero to -1 and zero to +3
D.zero to +1 and zero to -3
Solution
When chlorine gas reacts with hot and concentrated NaOH solution, it disproportionates into chloride (Cl- ) and chlorate (Cl3-) ions.
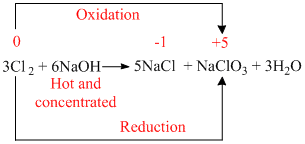
In this process, oxidation number of chorine changes from 0 to -1 and 0 to +5.
Note: In disproportionation reactions, the same element undergoes oxidation as well as reduction

In this process, oxidation number of chorine changes from 0 to -1 and 0 to +5.
Note: In disproportionation reactions, the same element undergoes oxidation as well as reduction
Create a free account to view solution
View Solution FreeMore Mole Concept Questions
16 g of an ideal gas SOx occupies 5.6 L. at STP. The value of x is...Mole fraction of the solute in a 1.00 molal aqueous solution is...Given that the abundances of isotopes 54Fe, 56Fe and 57Fe are 5%, 90% and 5% respectively, the atomic mass of Fe is...Consider the molecules CH4,NH3 and H2O. Which of the given statements is false?...One mole of the aquous solution of complex CoCl3.4NH3 passess through anion exchange resine and resulting solution treat...