Mole ConceptHard
Question
16 g of an ideal gas SOx occupies 5.6 L. at STP. The value of x is
Options
A.x = 3
B.x = 2
C.x = 4
D.none
Solution
Mol. wt. of gas is = 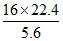 = 64 g
= 64 g
32 + 16x = 64
x = 2
32 + 16x = 64
x = 2
Create a free account to view solution
View Solution FreeMore Mole Concept Questions
Law of multiple proportions is not applicable for the oxide(s) of...Hydrogen cyanide, HCN, is prepared from ammonia, air and natural gas (CH4) by the following process. 2NH3(g) + 3O2(g) + ...When a sample of hydrogen fluoride is cooled to 303 K, most of the molecules undergo dimerization. If the vapour density...Silver metal in ore is dissolved by potassium cyanide solution in the presence of air by the reaction 4 Ag + 8 KCN + O2 ...When a mixture of aluminium powder and iron (III) oxide is ignited, it produces molten iron and aluminium oxide. In an e...