Mole ConceptHard
Question
Mole fraction of the solute in a 1.00 molal aqueous solution is
Options
A.0.0177
B.0.0344
C.1.7700
D.0.1770
Solution
1.00 molal aqueous slution = 1.0 mole in 1000 g water nSolute = 1; WSolvent = 1000 g.
nSolvent =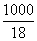 = 55.56
= 55.56
XSolute =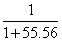 = 0.0177
= 0.0177
nSolvent =
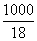 = 55.56
= 55.56 XSolute =
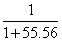 = 0.0177
= 0.0177Create a free account to view solution
View Solution FreeMore Mole Concept Questions
1M 100 ml NaCl is mixed with 3M 100 ml HCl solution and 1M 200 ml CaCl2 solution. Find the ratio of the concentration of...From 2 mg calcium, 1.2 × 1019 atoms are removed. The number of g-atoms of calcium left is (Ca = 40)...Elevation in boiling point of an aq. solution of urea is 0.52 (Kb for water = 0.52K molality-1). The mole fraction of ur...6.02 × 1020 molecules of urea are present in 100 ml of its solution. The concentration of solution is :-...A compound contains 7 carbon atoms, 2 oxygen atoms and 9.96 × 10–24 g of other elements. The molecular mass of compound ...