Mole ConceptHard
Question
Given that the abundances of isotopes 54Fe, 56Fe and 57Fe are 5%, 90% and 5% respectively, the atomic mass of Fe is
Options
A.55.85
B.55.95
C.55.75
D.56.05
Solution
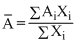
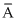 = 54 × 0.05 + 56 × 0.90 + 57 × 0.05 (where
= 54 × 0.05 + 56 × 0.90 + 57 × 0.05 (where 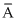 is atomic mass of Fe)
is atomic mass of Fe)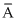 = 55.95
= 55.95Create a free account to view solution
View Solution FreeMore Mole Concept Questions
The molar concentration of HCl (aq.) is 10-5 M. Which of the following statements are correct.(dsolution = 1 gm /cc)...When 20 g Fe2O3 is reacted with 50 g of HCl, FeCl3 and H2O are formed. The amount of unreacted HCl is(Fe = 56)...In which of the following reactions, increase in the volume at constant temperature does not affect the number of moles ...In a textile mill, a double effect evaporator system concentrates weak liquor containing 4% (by mass) caustic soda to pr...The mass of 3.2 × 105 atoms of an element is 8.0 × 10−18 g. The atomic mass of the element is about(NA = 6 × 1023)...