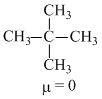Chemical BondingHard
Question
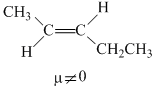
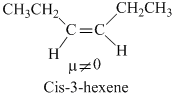
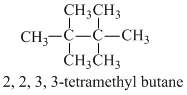
The molecules that will have dipole moment are
Options
A.2, 2-dimethyl propane
B.trans-2-pentene
C.cis-3-hexene
D.2, 2, 3, 3-tetramethyl butane
More Chemical Bonding Questions
The correct has high electron affinity ?...The correct statement(s) regarding, (i) HClO, (ii) HClO2, (iii) HClO3 and (iv) HClO4, is(are)...Which of the following bonds have lowest bond energy :-...Specify the coordination geometry around and hybridisation of N and B atoms in a 1 : 1 complex of BF3 and NH3 :...The total number of p-bond electrons in the following structure is :-...