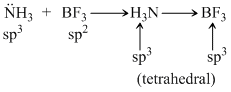Chemical BondingHard
Question
Specify the coordination geometry around and hybridisation of N and B atoms in a 1 : 1 complex of BF3 and NH3 :
Options
A.N : tetrahedral, sp3 ; B : tetrahedral, sp3
B.N : pyramidal, sp3 ; B : pyramidal, sp3
C.N : pyramidal, sp3 ; B : planar, sp2
D.N : pyramidal, sp3 ; B : tetrahedral, sp3
More Chemical Bonding Questions
Four diatomic species are listed below. Identify the correct order in which the bond order is increasing:...Which one of the following arrangements of molecules is correct on the basis of their dipole moments?...Select the correct option it is known thatKsp(AgCl) > Ksp (AgBr) > Ksp (AgI)...Identify the molecule $(X)$ with maximum number of lone pairs of electrons (obtained using Lewis’s dot structure) among ...Which of the following species contain three bond pairs and one lone pair around the central atom ?...