Chemical BondingHard
Question
The correct has high electron affinity ?
Options
A.Li+ > Na+ > K+ > Rb-
B.Rb+ > K+ > Na+ > Li+
C.Na+ > K+ > Li+ > Rb+
D.K+ > Rb+ > Na+ > Li+
Solution
hydration energy ∝ 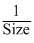
In a group, size increase on going down the group from top to bottom i.e.,
Rb+ > K+ > Na+ > Li+
∴ Increasing order of hydration energy Li+ Na+ > K+ > Rb+
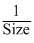
In a group, size increase on going down the group from top to bottom i.e.,
Rb+ > K+ > Na+ > Li+
∴ Increasing order of hydration energy Li+ Na+ > K+ > Rb+
Create a free account to view solution
View Solution FreeMore Chemical Bonding Questions
Which of following is not correctly matched ?...The charge/size ratio of a cation determines its polarizing power. Which one of the following sequences represents the i...In the reacdtion sequence2CH3CHO AB ; the product B is :...Both [Ni(CO)4] and [Ni(CN)4]2- are diamagnetic. The hybridization of nickel in these complexes, respectively, are...In a multistep reaction such as A + B → Q → C. The potential energy diagram is shown below. What is Ea for t...