AIPMT | 2015Chemical BondingHard
Question
The total number of p-bond electrons in the following structure is :-
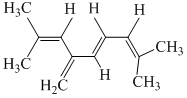

Options
A.8
B.12
C.16
D.4
Solution
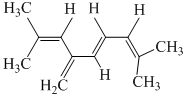
Total π bonds = 4
Total π electrons = 8
Create a free account to view solution
View Solution FreeMore Chemical Bonding Questions
Match the following Ore Chemical Formula(A) Limonite (i) Fe2O3.3H2O(B) Cryolite (ii) Cu(OH)2.2CuCO3(C) Calamine (iii) Na...Which of the following structure is similar to graphite?...In which pair of species the hybrid state of B is sp3 :-...Which of the following statements is/are correct ?...Correct increasing order for the wavelengths of absorption in the visible region the complexes of Co3+ is :-...