JEE Advanced | 2015Chemical BondingHard
Question
The correct statement(s) regarding, (i) HClO, (ii) HClO2, (iii) HClO3 and (iv) HClO4, is(are)
Options
A.The number of Cl = O bonds in (ii) and (iii) together is two
B.The number of lone pairs of electrons on Cl in (ii) and (iii) together is three
C.The hybridization of Cl in (iv) is sp3
D.Amongst (i) to (iv), the strongest acid is (i)
Solution
Structure of
(i) HClO =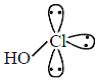
(ii) HClO2 =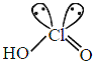
(iii) HClO3 =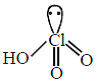
(iv) HClO4 =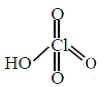
(A) The number of Cl = O bonds in (ii) and (iii) together is three
(B) The number of lone pairs of electrons on Cl in (ii) and (iii) together is three
(C) The hybridisation of Cl in (iv) is sp3
(D) Amongst (i) to (iv) the strongest acid is (iv) HClO4
(i) HClO =
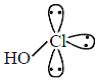
(ii) HClO2 =
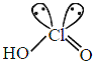
(iii) HClO3 =
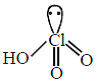
(iv) HClO4 =

(A) The number of Cl = O bonds in (ii) and (iii) together is three
(B) The number of lone pairs of electrons on Cl in (ii) and (iii) together is three
(C) The hybridisation of Cl in (iv) is sp3
(D) Amongst (i) to (iv) the strongest acid is (iv) HClO4
Create a free account to view solution
View Solution FreeMore Chemical Bonding Questions
The major product in the following reaction is...Given below are two statements:Statement I: Among $\left\lbrack Cu\left( {NH}_{3} \right)^{2 +},\left\lbrack Ni(en)_{3} ...In which of the following pairs of molecules/ions, both the species are not likely to exist ?...Which of the following statements is/are correct ?Commercial HCl is prepared by heating NaCl with H2SO4.2NaCl + H2SO4 &#...In which of the folloiwng species the interatomic bond angle is 109o28′ ?...