Chemical EquilibriumHard
Question
The value of Kp for the reaction, 2H2O(g) + 2Cl2(g) ⇋ 4HCl (g) + O2 (g)
is 0.03 atm at 427oC, when the partial pressure are expressed in atmosphere then the value of KC for the same reaction is
is 0.03 atm at 427oC, when the partial pressure are expressed in atmosphere then the value of KC for the same reaction is
Options
A.5.23 × 10-4
B.7.34 × 10-4
C.3.2 × 10-3
D.5.43 × 10-5
Solution
2H2O (g) + 2Cl2 (g) ⇋ 2HCl (g) + O2 (g)
Kp = 0.03 T = 477oC = 700 K
Kp = KC(RT)1
KC =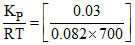
KC = 5.23 × 10-4
Kp = 0.03 T = 477oC = 700 K
Kp = KC(RT)1
KC =
KC = 5.23 × 10-4
Create a free account to view solution
View Solution FreeMore Chemical Equilibrium Questions
Ammonia gas at 15 atm is introduced in a rigid vessel at 300 K. At equilibrium the total pressure of the vessel is found...Under what pressure must an equimolar mixture of Cl2 and PCl3 be placed at 250oC in order to obtain 75% conversion of PC...An amount of 1 mole each of A and D is introduced in 1 L container. Simultaneously the following two equilibria are esta...A vessel at 1000 K contains CO2 with a pressure of 0.5 atm. Some of the CO2 is converted into CO on the addition of grap...The equilibrium mixture for the reaction 2H2S(g)$\rightleftharpoons$ 2H2(g) + S2(g) has 1 mole of H2S, 0.20 mole of H2 a...