Chemical EquilibriumHard
Question
A vessel at 1000 K contains CO2 with a pressure of 0.5 atm. Some of the CO2 is converted into CO on the addition of graphite. If the total pressure at equilibrium is 0.8 atm, the value of K is
Options
A.3 atm
B.0.3 atm
C.0.18 atm
D.1.8 atm
Solution
CO2(g) + C → 2CO(g)
Initial moles p O
Equilibriumm moles p-x 2x
Total pressure at equilibrium = 0.8 atm ; Total no.of moles = p + x.
Therefore p ∝ n;
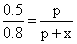 ⇒ x = 0.3
⇒ x = 0.3Kp =
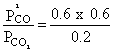 = 1.8 atm
= 1.8 atmCreate a free account to view solution
View Solution FreeMore Chemical Equilibrium Questions
In the system AB(s) $\rightleftharpoons$A(g) + B(g), doubling the quantity of AB(s) would...The value of KP for the process: CuSO4·5H2O(s) $\rightleftharpoons$CuSO4·3H2O(s) + 2H2O(g) is 1.21 × 10–4 atm2 at certai...Forty percent of a mixture of 0.2 mol of N2 and 0.6 mol of H2 reacts to give NH3 according to the equationN2(g) + 3H2 (g...When two reactants, A and B are mixed to give products C and D, the reaction quotient Q at the initial stages of the rea...NH4HS(S) ⇋ NH3(g) + H2S(g)The equilibrium pressure at 25oC is 0.660 atm. What is KP for the reaction....