Chemical EquilibriumHard
Question
Ammonia gas at 15 atm is introduced in a rigid vessel at 300 K. At equilibrium the total pressure of the vessel is found to be 40.11 atm at 300oC. The degree of dissociation of NH3 will be:
Options
A.0.6
B.0.4
C.Unpredictable
D.None of these
Solution
P1 = 15 atm ; T1 = 300 K.
Equilibrium temperature is 300oC that is 573 K.
So first of all we have to calculate pressure of NH3 at 573 K.
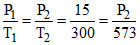
P2 = 28.65 atm at 300oC.
NH3 (g) ⇋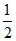 N2(g) +
N2(g) + 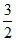 H2(g).
H2(g).
t = 0 28.65 atm 0 0
t = teq. [28.65-x]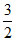 atm
atm 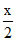 x
x
But according to question.
Ptotal = 28.65 - x +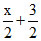 x
x
= 28.65 = x = 40.11.
x = 11.46.
Degree of dissociation of NH3 =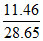 = 0.4.
= 0.4.
Equilibrium temperature is 300oC that is 573 K.
So first of all we have to calculate pressure of NH3 at 573 K.
P2 = 28.65 atm at 300oC.
NH3 (g) ⇋
t = 0 28.65 atm 0 0
t = teq. [28.65-x]
But according to question.
Ptotal = 28.65 - x +
= 28.65 = x = 40.11.
x = 11.46.
Degree of dissociation of NH3 =
Create a free account to view solution
View Solution FreeMore Chemical Equilibrium Questions
For the equilibrium A(g) $\rightleftharpoons$nB(g), the equilibrium constant KP is related with the degree of dissociati...KMnO4 can be prepared from K2MnO4 as per the reaction :- 3MnO42- + 2H2O ⇋ 2MnO4- + MnO4 + 4OH-The reaction can go ...CuSO4.5H2O(s) $\rightleftharpoons$CuSO4.3H2O(s) + 2H2O(g), KP for this equilibrium is 1.0 × 10−4 atm2 at 25oC. What is t...Iron fillings and water were placed in a 5 L vessel and sealed. The tank was heated to 1000oC. Upon analysis, the tank w...For the following gases equilibrium, N2O4 (g) ⇋ 2NO2 (g)Kp is found to be equal to Kc. This is attained when :...