Atomic StructureHard
Question
The energy of an electron in the first Bohr orbit of H-atom is -13.6eV. The possible energy value (s) of the excited state (s) for electrons in Bohr orbits of hydrogen is (are) :
Options
A.- 3.4 eV
B.- 4.2eV
C.- 6.8eV
D.+ 6.8 eV
More Atomic Structure Questions
The radii of two of the first four Bohr′s orbits of the hydrogen atom are in the ratio 1 : 4 The energy difference...Cumbrous ion is collarless while cupric ion is colored because...If E1, E2 and E3 are the kinetic energies of an electron, an α-particle and a proton with the same de Broglie wavelength...Photon having wavelength 310 nm is used to break the bond of A2 molecule having bond energy 288 kJ mol-1 then % of energ...The magnitude of the orbital angular momentum of an electron is given by $L = \sqrt{5}h/\pi$. How many orbitals of this ...
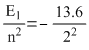 = - 3.4eV
= - 3.4eV