Atomic StructureHard
Question
Photon having wavelength 310 nm is used to break the bond of A2 molecule having bond energy
288 kJ mol-1 then % of energy of photon converted to the K.E. is [hc = 12400 ev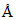 , 1 ev = 96 kJ/mol]
, 1 ev = 96 kJ/mol]
288 kJ mol-1 then % of energy of photon converted to the K.E. is [hc = 12400 ev
Options
A.25
B.50
C.75
D.80
Solution
Energy of one photon = 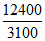 = 4ev = 4 × 96 = 384 kJ mol-1
= 4ev = 4 × 96 = 384 kJ mol-1
∴ % of energy converted to K.E. =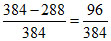 × 100 = 25%
× 100 = 25%
∴ % of energy converted to K.E. =
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
The dissociation energy of H2 is 429.0 kJ/mol. If H2 is dissociated by illumination with radiation of wavelength 270.0 n...The radii of two of the first four Bohr′s orbits of the hydrogen atom are in the ratio 1 : 4 The energy difference...The speed of electron revolving in the fourth orbit of a hydrogen-like atom or ion is 1094 km/s. The atom or ion is...An electron makes five crests during one revolution around H-nucleus. The electron belongs from the...Hyperconjugation involves overlap of the following orbitals...