Chemical EquilibriumHard
Question
Rate of reaction curve for equilibrium can be like : [rf = forward rate, rb = backward rate]
Options
A.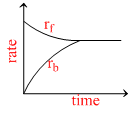

B.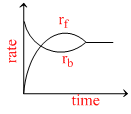

C.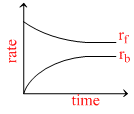

D.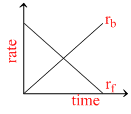

Solution
At equilibrium rates of backward and forward reactions become equal.
Create a free account to view solution
View Solution FreeMore Chemical Equilibrium Questions
In the dissociation of N2O4 into NO2, (1 + α) values with the vapour densities ratio is as given by : [a-degree of ...At 0°C and 1 atm pressure, 1 L of N2O4 decomposes to NO2 according to the equation N2O4(g) $\rightleftharpoons$2NO2 (g)....Solid NH4HS dissociates into NH3 and H2S at a certain temperature, the equilibrium pressure is P atm. If now, NH3 is pum...The value of Kp for the reaction, 2H2O(g) + 2Cl2(g) ⇋ 4HCl (g) + O2 (g)is 0.03 atm at 427oC, when the partial pres...When S in the form of S8 is heated at 900 K, the initial pressure of 1 atm falls by 30% at equilibrium. This is because ...