Chemical EquilibriumHard
Question
In the dissociation of N2O4 into NO2, (1 + α) values with the vapour densities ratio 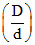 is as given by : [a-degree of dissociation, D-vapour density before dissociation, d-vapour density after dissociation]
is as given by : [a-degree of dissociation, D-vapour density before dissociation, d-vapour density after dissociation]
Options
A.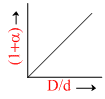

B.

C.

D.

Solution
N2O4 ⇋ 2NO2 Mmix = 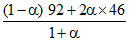
1 - α 2α Mmix =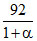
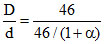 = (1 + α). Therefore, (A) option is correct.
= (1 + α). Therefore, (A) option is correct.
1 - α 2α Mmix =
Create a free account to view solution
View Solution FreeMore Chemical Equilibrium Questions
1 mol of N2 is mixed with 3 mol of H2 in a litre container. If 50% of N2 is converted into ammonia by the reaction N2 + ...For the reaction CaCO3(s) $\rightleftharpoons$CaO(s) + CO2(g), the value of KP is...For the reaction CaCO3(s) ⇋ CaO(s) + CO2(g), the pressure of CO2(g) depends on :...When alcohol (C2H5OH (l)) and acetic acid (CH3COOH (l)) are mixed together in equimolar ratio at 27oC , 33% of each is c...The Haber’s process for the manufacture of ammonia is usually carried out at about 500oC. If a temperature of about 250o...