SolutionHard
Question
Calculate the osmotic pressure of the solution prepared in the above question T = 300 K, (R = 0.082 L atm mol-1K-1)
Options
A.10.8 atm
B.12.8 atm
C.5.6 atm
D.None of these
Solution
π = CRT
C = (0.34 +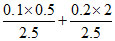 ) = 0.34 + 0.02 + 0.16 = 0.52
) = 0.34 + 0.02 + 0.16 = 0.52
so π = 0.52 × 0.082 × 300 atm = 12.792 atm
C = (0.34 +
so π = 0.52 × 0.082 × 300 atm = 12.792 atm
Create a free account to view solution
View Solution FreeMore Solution Questions
Observe the P-T phase diagram for a given substance A. Then melting point of A(s), boiling point of A(l), critical point...Select incorrect statement :...Two liquids A and B form an ideal solution at temperature T K. At T K, the vapour pressures of pure A and B are 55 and $...The fraction of phenol dimerised in benzene if 20 g of phenol in 1 kg benzene exhibits a freezing point depression of 0....An aqueous solution of a solute AB has b.p. of 101.08oC (AB is 100% ionised at boiling point of the solution) and freeze...